The Haber-Bosch process, which produces ammonia by reacting nitrogen and hydrogen gases in the presence of an iron catalyst at high pressure and temperature, is used to generate ammonia in industrial settings. This highly energy-intensive method must carefully manage the reaction conditions to attain the best yield.
Introduction
Most people associate ammonia with the pungent smell of cleaners and salts. Ammonia (Chemical formula - NHз) is one of the most common chemicals known to humankind. It is convenient and used to make other valuable products, such as fertilisers and polymers.
Large-scale ammonia gas manufacturing occurs in massive facilities or plants. Ammonia has been produced yearly since 1946, and its production is expected to reach 150 million tonnes in 2022. This makes it one of the most synthetically produced chemicals in the world. China and Russia are currently the two countries with the highest annual production rates.
Ammonia and goods containing ammonia have dramatically increased due to the growing population. This has caused various changes to ammonia production procedures. Let's see how modern industry has evolved.
Do you know?
The amount of ammonia that can be manufactured annually worldwide was capped at 235 million tonnes in 2021, the highest level to date. Also, 80% of the ammonia produced is used to make fertilisers.
Ammonia - Importance and Uses
Ammonia is considered the building block of many compounds required to produce essential products.
- Nitrogen-based Fertilisers: Ammonia is one of the most accessible nitrogen sources. Thus, around 80% of the ammonia produced is used to produce nitrogen-based fertilisers such as Urea, as nitrogen is vital for plant growth.
- Refrigeration and Plastic Manufacturing: Apart from fertilisers, ammonia is also used for refrigeration and manufacturing plastics, synthetic fibres, dyes, and other valuable chemicals.
- Wastewater Treatment and Purification: It is also used in wastewater treatment and purification, in the cosmetic industry and in producing certain pharmaceuticals.
Also Read: How Are Antibiotics Made? The Manufacturing Process Explained
History of Ammonia Production
Ammonia production dates back to the pre-World War 1 era. Even though many methods existed to produce ammonia in the lab, like the Cynamide method developed by Adolph Frank and Nikodem Caro, they were too inefficient. Thus the era lacked a process to produce ammonia economically.
Fritz Haber and Carl Bosch, two famous German scientists, developed the first methods to produce ammonia on an industrial scale. Their process was named after them in their honour and is known as the ‘Haber-Bosch’ process.
Haber-Bosch was developed and first used in the 20th century. The process included the production of ammonia with atmospheric nitrogen by a reaction with hydrogen under a specified set of conditions like pre-defined temperature, pressure and the presence of a metal catalyst.
The hydrogen required for the procedure is obtained via steam reforming. This hydrogen is then reacted with nitrogen to produce Ammonia (NHз).
The first ammonia production plant was opened in Oppau, Germany, on the 19th of September 1913, with a production capacity of 30 metric tonnes per day.
Ammonia Production - Conventional Methods
Most conventional methods are based on the popular Haber-Bosch process. The nitrogen and hydrogen required for the procedure are produced using different methods:
1. By Coal
In Asian countries like China or Russia, coal is the primary source of energy production. The energy required for ammonia synthesis is supplied by coal burning. Coal is converted to coal gas and further synthesised to produce ammonia.
2. By Natural Gas
In this method, methane or natural gas reacts with steam and air to produce Hydrogen, Nitrogen and CO2, and H2O as a by-product. Hydrogen and nitrogen are used as supplies for ammonia production.
The main downside is the production of impurities like argon which damages machinery. Thus, it requires a separate filtration and condensation plant to separate ammonia from its byproducts.
3. By Water-electrolysis and Air Separation
In this method, the nitrogen required is obtained via air separation, and the hydrogen required is produced by conducting water electrolysis. Air separation of nitrogen is performed by cryogenic absorption or membrane or adsorption processes.
This method is much more economical to produce ammonia on an industrial scale than the above-mentioned processes.

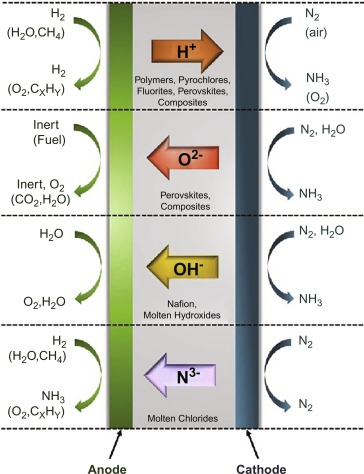
Ammonia Production by Electrochemical Method
This ammonia synthesis method is much simpler and easier to execute than the systems based on the Haber-Bosch process. Also, the process has better energy utilisation as it uses about 20% less energy than previous methods.
This method includes a simple cathode, anode and electrolyte layer setup. Hydrogen is supplied to the anode, and nitrogen is supplied to the cathode.
Due to the electrolysis process, nitrogen at the cathode reacts with hydrogen ions released at the anode to give ammonia. Since the process is straightforward to execute, it reduces machinery investment and maintenance costs and reduces complexity in the system.
Ammonia production cost dates back to the pre-World War 1 era. Even though many methods existed to produce ammonia in the lab, like the Cynamide method developed by Adolph Frank and Nikodem Caro, they were too inefficient. Thus the era lacked a process to produce ammonia economically.
The procedure's efficiency depends on the type of electrolyte used.
Also Read: How Are Carbon Papers Made - The Mayer and Flexographic Method Explained
Currently, four types of electrolytes are available in the market: liquid, molten, composite and solid.
1. Liquid electrolytes like LiClO4(0.2 M in tetrahydrofuran) can easily operate at room temperatures and pressure, providing around 58-60% efficiency.
2. Molten electrolytes like LiCl or KCl dissolved in Li3N can operate at temperatures ranging from 300-700°C and provide an efficiency rate of 72-74%.
3. Composite electrolytes like LiCO3, LiAlO2 and CeO2 doped with Sm2O3 include an ionic conducting phase and an additional phase sandwiched with the main layer. This is to improve the chemical and thermal properties of the electrolyte.
These types of electrolytes operate at temperatures around 400°C.
4. Solid-state electrolytes can use perovskites, fluorites or pyrochlores. These electrolytes can operate at a wide range of temperatures ranging from room temperatures to 800°C, providing an efficiency rate of 75%.
However, a downside of these electrolytes is that they have low structural stability and a very high sintering temperature (around 1700°C).
Ammonia Production by the Thermochemical Cycle
This production method includes two significant steps: reduction (activation of nitrogen) and steam-hydrolysis (ammonia production).
Unlike other processes, this method does not require any special conditions or the presence of any catalysts and can be easily carried out at atmospheric pressure, making it convenient.
Also, this method does not require pure hydrogen and can use a carbonised method to reduce energy consumption. The system is operated under the necessary conditions and can provide up to 70% efficiency.
The biggest downside of this process is the operating temperature which is very high compared to other processes. This makes the process very limited.

Modern Methods of Ammonia Production
Modern ammonia production includes a purifier process. Due to increasing environmental consciousness, production processes have become energy-efficient and environmentally friendly.
1. M.W. Kellog, better known as MWK, brought the first revolution in ammonia production after Haber and Bosch.
Their Texas City ammonia plant is well known. The plant became popular because of a single train design which gave it a proper balance between energy consumption and ammonia production rates. The idea of a single train system was revolutionary then, and it helped the team secure several awards for them.
The plant was one of the leading ammonia producers in the 1960s, with a capacity of 544 metric tonnes per day.
2. Newer ammonia production industries mostly have plants based on technologies developed by KBR (Kellog Brown and Root), Ammonia Casale, etc.
-
Plants based on KBR technology include a primary reformer, a liquid nitrogen wash purifier, a heating boiler, a unitised chiller, and an ammonia synthesis converter. These plants are incredibly efficient and can consume energy as low as 28 GJ/metric ton.
-
The primary reformers used in these plants are way smaller than conventional designs. This helps to reduce excess air usage and transfer the load to secondary reformers.
-
The cryogenic purifier chambers comprise a condenser, deed, effluent exchange column and rectifier column. It removes any kind of atomic level impurities like CH4 or carbon monoxide and other impurities from the synthesis gas. Also, it helps to maintain the nitrogen: hydrogen ratio in the makeup gas.
-
Plants based on Ammonia Casale’s technology include an extraordinary axial-radial tech in the catalyst bed. This helps create a very low-pressure drop throughout the process and makes the plant usable at both high and low temperatures. Such plants can produce up to 2000 metric tonnes per day.
Conclusion
Ammonia is a crucial compound in our daily lives. From playing an essential role in the production of green vegetables to the refrigeration required to maintain those vegetables, from the medicines we take to the clothes we wear, we all need ammonia for production at some point.
Ammonia production methods have changed dramatically in the past few decades, and production processes have become more efficient and environmentally friendly.
Fritz Haber and Carl Bosch did the earliest development in ammonia production. This was further carried out by companies like M.W. Kellogg, Ammonia Casale, KBR, etc.
Follow Khatabook for the latest updates, news blogs, and articles related to micro, small and medium businesses (MSMEs), business tips, income tax, GST, salary, and accounting.

